Oxidation and Reduction of Carbonyl Compounds
Important Questions on Oxidation and Reduction of Carbonyl Compounds
Write the product of following reaction
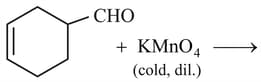
Write the product of following reaction:
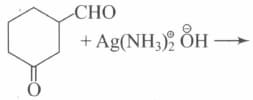
Which is/are correct statement?
In which of the following cases, products formed are not according to the reaction?
The suitable reagent of the following reaction is:
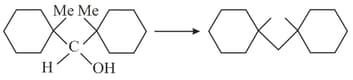
A compound (A) forms a phenyl hydrazone and gives negative Tollen's and positive iodoform test compound can be:
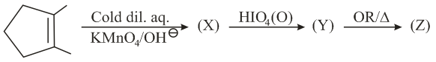
The products of the above reaction is/are:
Following is chemoselective for the reduction of carbonyl compounds to corresponding alcohol:
Which of the following does not turn Schiff's reagent to pink?
is oxidised to by using
Acetaldehyde on warming with Fehling's solution gives a red precipitate of
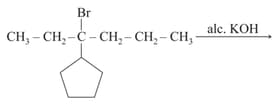 (mixture of alkenes products) (mixture of carbonyl compounds)
(mixture of alkenes products) (mixture of carbonyl compounds)
The incorrect statement is:
Compound gives a yellow coloured precipitate with , but does not give red coloured precipitate with Fehling's solution. on treatment with gives a compound . , when treated with , gives an isomeric compound , on hydrolysis gives propanoic acid and aniline. What will be the correct structure of , and ?
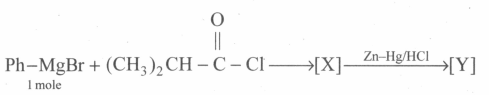
Identify the structure of [Y].
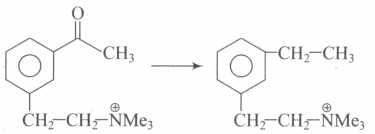 Above conversion can be achieved by:
Above conversion can be achieved by:
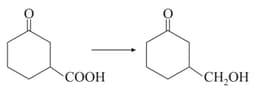
Show how would you accomplish the following synthesis:
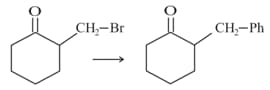
How would you accomplish the following synthesis?
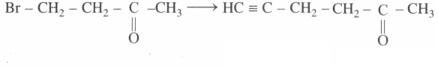
How would you accomplish the following synthesis?


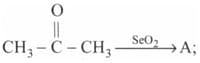
 and are :
and are :