MEDIUM
Earn 100
A fixed mass of air at atmospheric pressure is compressed adiabatically to atmospheres and allowed to expand isothermally to its original volume. The process is represented in which one of the following indicator diagrams?
(a)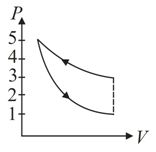

(b)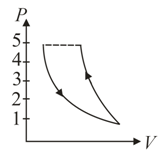

(c)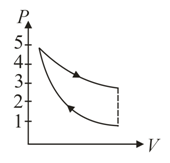

(d)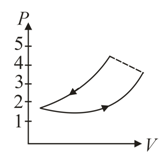

50% studentsanswered this correctly
Important Questions on Thermodynamics
EASY
One mole of an ideal diatomic gas undergoes a transition from to along a path as shown in the figure,
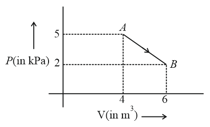
The change in internal energy of the gas during the transition is:
EASY
MEDIUM
One mole of diatomic ideal gas undergoes a cyclic process ABC as shown in figure. The process BC is adiabatic. The temperatures at A, B and C are 400 K, 800 K and 600 K respectively. Choose the correct statement :
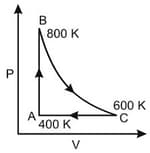
EASY
EASY
HARD
EASY
(Take gas constant )
MEDIUM
MEDIUM
MEDIUM
EASY
EASY
EASY
MEDIUM
An ideal gas is taken reversibly around the cycle as shown on the T (temperature) - S (entropy) diagram
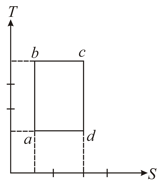
The most appropriate representation of above cycle on a U (internal energy)-V (volume) diagram is
HARD
(Take , where is gas constant)
EASY
Thermodynamic processes are indicated in the following diagram.
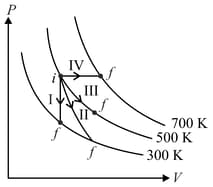
Match the following
| Column – 1 | Column - 2 |
| P. Process I | a. Adiabatic |
| Q. Process II | b. Isobaric |
| R. Process III | c. Isochoric |
| S. Process IV | d. Isothermal |
MEDIUM
EASY
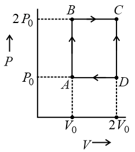
For the given cyclic process as shown for a gas, the work done is:
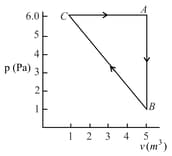
EASY
HARD

