A gas is made to undergo a change of state from an initial state to a final state along different paths by adiabatic process only. Therefore.

Important Questions on Thermodynamics
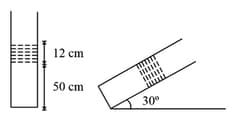
In the given figure, an ideal gas is trapped between a mercury column and the closed end of a uniform vertical tube. The upper end of the tube is open to the atmosphere. Initially, the lengths of the mercury column and the trapped air column are and , respectively. When the tube is tilted slowly in a vertical plane through an angle of with horizontal, then the new length of air column is . Find (assume the temperature to remain constant ).
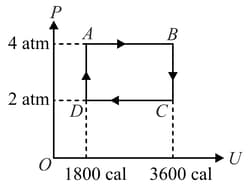
Two moles of an ideal monoatomic gas undergo a cyclic process which is indicated on a diagram where is the internal energy of the gas. The work done by the gas in the cycle is . Find .
A cyclic process is shown in the P–V diagram. ( and are isothermal)
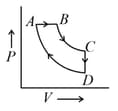
Which of the following curves represent the same process?
