EASY
Earn 100
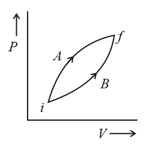
A gas undergoes change from state to state . In this process, the heat absorbed and work done by the gas is , respectively. Now gas is brought back to by another process during which of heat is evolved. In this reverse process of .
(a)of the work will be done by the gas
(b) of the work will be done by the surrounding on gas
(c) of the work will be done by the surrounding on gas
(d) of the work will be done by the gas
(e) of the work will be done by the surrounding on gas
50% studentsanswered this correctly
Important Questions on Chemical Thermodynamics and Energetics
EASY
EASY
MEDIUM
MEDIUM
(R = 8.314 J/mol K) (ln7.5 = 2.01)
EASY
MEDIUM
HARD
The specific heat of a certain substance is . Assuming ideal solution behavior, the energy required (in ) to heat of molal of its aqueous solution from to is closest to :
[Given: molar mass of the substance ; specific heat of water ]
EASY
MEDIUM
EASY
MEDIUM
MEDIUM
EASY
MEDIUM
MEDIUM
of nitrous oxide gas is cooled at a constant pressure of atm from to causing the compression of the gas from to . The change in internal energy of the process, is . The value of is _____.
[nearest integer]
(Given: atomic mass of and of . Molar heat capacity of is )
EASY
EASY
EASY
HARD
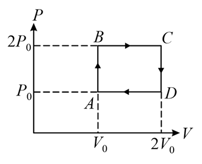
The above diagram represents the thermodynamic cycle of an engine, operating with an ideal mono-atomic gas. The amount of heat, extracted from the source in a single cycle, is:

