HARD
Earn 100
A student prepares 2 L buffer solution of 0.3 M NaH2PO4 and 0.3 M Na2HPO4. The solution is divided in half between the two compartments (each containing 1 L buffer) of an electrolysis cell, using Pt electrodes. Assume that the only reaction is the electrolysis of water and the electrolysis is carried out for 200 min with a constant current of 0.965 A.
[Given log 2.33 = 0.37]
pH at anode is
[Given log 2.33 = 0.37]
(a)
(b)
(c)
(d)
50% studentsanswered this correctly
Important Questions on Equilibrium
EASY
HARD
MEDIUM
Which of the following mixtures will have the lowest pH at
EASY
MEDIUM
[Given:
MEDIUM
MEDIUM
HARD
[Given: of acetic acid molar mass of acetic acid , Neglect any changes in volume]
MEDIUM
MEDIUM
EASY
HARD
HARD
MEDIUM
EASY
EASY
HARD
A solution of weak base is titrated with of a strong acid . The variation of of the solution with the volume of added is shown in the figure below. What is the of the base? The neutralisation reaction is given by .
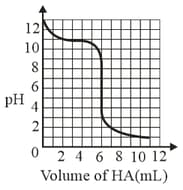
MEDIUM
EASY

