A vessel contains mole of gas (molar mass ) at a temperature . The pressure of the gas is . An identical vessel containing one mole of He gas (molar mass ) at temperature has a pressure of

Important Questions on Thermometry, Thermal Expansion and Kinetic Theory of Gases
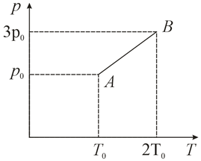
Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point is . Density at will be,
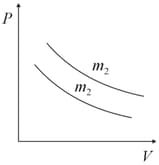
Two different isotherms representing the relationship between pressure and volume at a given temperature of the same ideal gas are shown for masses and , then
Each atom of mass of a monoatomic gas has got three degrees of freedom. The velocity of these atoms is at temperature For a diatomic molecule of mass and temperature , which has got five degrees of freedom, rms velocity of molecule is,
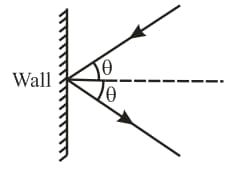
The mass of hydrogen molecule is . If hydrogen molecules strike per second at area of a rigid wall at an angle of from the normal and rebound back with a speed of , then the pressure exerted on the wall is,
A ring shaped tube contains two ideal gases with equal masses and relative molar masses and . The gases are separated by one fixed partition and another movable stopper , which can move freely without friction inside the ring.
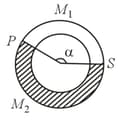
The angle in equilibrium as shown in the figure (in degrees) is
