HARD
Earn 100
Assertion: The ratio for a diatomic gas is more than that for a monoatomic gas.
Reason: The molecules of a monoatomic gas have less degree of freedom than those of diatomic gas.
(a)If both Assertion and Reason are true and the Reason is correct explanation of the Assertion.
(b)If both Assertion and Reason are true but Reason is not correct explanation of the Assertion.
(c)If Assertion is true but the Reason is false.
(d)If Assertion is false but Reason is true.
18.18% studentsanswered this correctly
Important Questions on Thermodynamics
MEDIUM
HARD
MEDIUM
MEDIUM
What will be the molar specific heat at constant volume of an ideal gas consisting of rigid diatomic molecules?
EASY
EASY
HARD
MEDIUM
EASY
MEDIUM
MEDIUM
MEDIUM
HARD
EASY
EASY
EASY
MEDIUM
Two different metal bodies and of equal mass are heated at a uniform rate under similar conditions. The variation of temperature of the bodies is graphically represented as shown in the figure. The ratio of specific heat capacities is:
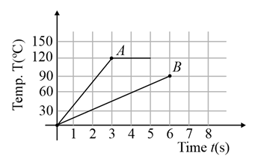
MEDIUM
[Given that
EASY
EASY
(Take gas constant )

