MEDIUM
JEE Main/Advance
IMPORTANT
Earn 100
Choose the correct statement:
(a)system and surrounding are always separated by a real or imaginary boundary.
(b)perfectly isolated system can never be created.
(c)in reversible process, energy change in each step can be reversed
(d)irreversible process is also called quasi-equilibrium state.
50% studentsanswered this correctly

Important Questions on Thermodynamics
MEDIUM
JEE Main/Advance
IMPORTANT
In an isothermal expansion of a gaseous sample, the correct relation is: (consider (work) with sign according to new IUPAC convention)
[The reversible and irreversible processes are carried out between same initial and final states.]
MEDIUM
JEE Main/Advance
IMPORTANT
MEDIUM
JEE Main/Advance
IMPORTANT
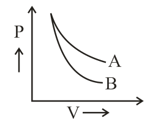
plot for two gases (assuming ideal) during adiabatic processes are given in the figure. Plot and plot should correspond respectively to:
MEDIUM
JEE Main/Advance
IMPORTANT
EASY
JEE Main/Advance
IMPORTANT
MEDIUM
JEE Main/Advance
IMPORTANT
Select the correct statements:
For every chemical reaction at equilibrium, the standard Gibbs energy of reaction is zero.
At constant temperature and pressure, chemical reactions are spontaneous in the direction of decreasing Gibbs energy.
Spontaneity is related to change in entropy of universe.
MEDIUM
JEE Main/Advance
IMPORTANT
MEDIUM
JEE Main/Advance
IMPORTANT
