EASY
Earn 100
Different gases at the same temperature have same
(a)Pressure
(b)Number of moles
(c)Volume
(d)Average kinetic energy
50% studentsanswered this correctly
Important Questions on Heat and Thermodynamics
EASY
EASY
EASY
MEDIUM
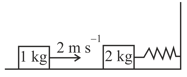
EASY
EASY
EASY
EASY
EASY
MEDIUM
EASY
EASY
EASY
EASY
EASY
EASY
EASY
At what temperature is the velocity of Hydrogen molecule is equal to that of an oxygen molecule at ?
(Molecular weight of hydrogen Molecular weight of oxygen )
EASY
EASY
EASY

