In a market with a fixed number of firms, if the market price is above the equilibrium price, what would be the result?
Important Questions on Equilibrium
A solid kept in an evacuated sealed container undergoes decomposition to form a mixture of gases at temperature .
The equilibrium pressure is in this vessel. for this reaction is?
When and are compared at It is found that
For the given reaction, if the initial pressure is and the pressure at time is at a constant temperature and constant volume . The fraction of decomposed under these conditions is . The value of is (nearest integer)
.....(1)
.....(2)
The relation between and is:
For a reaction at equilibrium
the relation between dissociation constant , degree of dissociation and equilibrium pressure is given by :
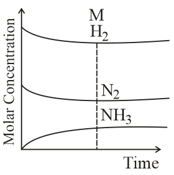
For the reaction
if the initial concentration of and moles of is consumed at equilibrium, the correct expression of is.
observed pressure for the reaction mixture in equilibrium is at . What is the value of for the reaction?
Which of the following statements is correct?
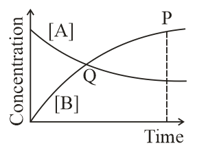
Among the following points indicated on the graph, which one represents equilibrium?