MEDIUM
Earn 100
In the pressure vs. the mole fraction of benzene curves/lines shown below, the total vapour pressure of an ideal mixture of benzene and toluene will follow the curve/line.
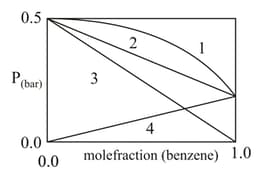

(a)
(b)
(c)
(d)
50% studentsanswered this correctly
Important Questions on Solutions
MEDIUM
MEDIUM
HARD
Benzene and toluene form an ideal solution over the entire range of composition. The vapour pressure of pure benzene and toluene at are and , respectively. What is the mole fraction of toluene in vapour phase when of benzene is mixed with of toluene?
(Molar mass of benzene and toluene are and , respectively)
EASY
MEDIUM
(
)
EASY
MEDIUM
MEDIUM
HARD
EASY
EASY
HARD
EASY
The correct option for the value of vapour pressure of a solution at with benzene to octane in molar ratio is:
[At vapour pressure of benzene is and that of octane is Assume Ideal gas]
HARD
HARD
(Given that the vapour pressure of pure liquid A is at temperature )
MEDIUM
MEDIUM
MEDIUM
MEDIUM
MEDIUM

