MEDIUM
Earn 100
Solubility of calcium phosphate (molecular mass, M) in water is W g per 100 mL at C. Its solubility product at will be approximately
(a)
(b)
(c)
(d)
50% studentsanswered this correctly
Important Questions on Ionic Equilibria
HARD
EASY
HARD
MEDIUM
HARD
MEDIUM
MEDIUM
MEDIUM
EASY
HARD
MEDIUM
MEDIUM
[Solubility product for ]
MEDIUM
Which of the following choices is correct for a mixture of and
MEDIUM
MEDIUM
The stoichiometry and solubility product of a salt with the solubility curve given below is, respectively:
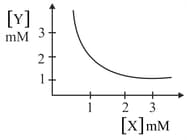
EASY
EASY
MEDIUM
HARD
MEDIUM

