MEDIUM
Earn 100
The correct order of the viscosity in the following compounds in liquid state is:
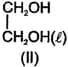
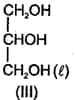
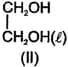
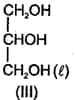
(a)
(b)
(c)
(d)
50% studentsanswered this correctly
Important Questions on States of Matter: Gases and Liquids
MEDIUM
The combination of plots which does not represent isothermal expansion of an ideal gas is
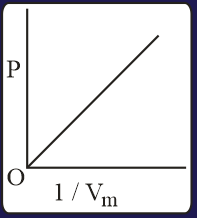
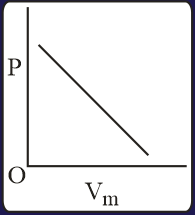
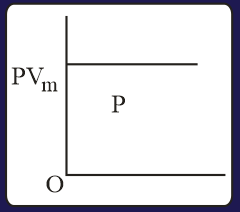
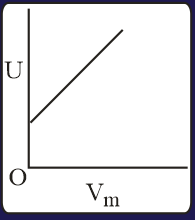
MEDIUM
The qualitative sketches I, II and III given below show the variation of surface tension with molar concentration of three different aqueous solutions of KCl, and at room temperature. The correct assignment of the sketches is -
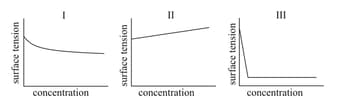
HARD
An ideal gas undergoes isothermal compression from to against a constant external pressure of . The heat released in this process is and is used to increase the pressure of mole of . The temperature of increases by:
MEDIUM
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of . As it does so, it absorbs 208 J of heat. The values of q and w for the process will be
(R = 8.314 J/mol K) (ln7.5 = 2.01)
MEDIUM
The units of surface tension and viscosity of a liquid, respectively, are
HARD
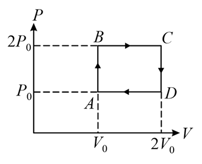
The above diagram represents the thermodynamic cycle of an engine, operating with an ideal mono-atomic gas. The amount of heat, extracted from the source in a single cycle, is:
EASY
A piece of ice falls from a height so that it melts completely. Only one-quarter of the heat produced is absorbed by the ice and all energy of ice gets converted into heat during its fall. The value of is:
(Latent heat of ice is and )
MEDIUM
of ice at is added to 340g of water at . The final temperature of the resultant mixture is . The value of (in g) is closest to
[Heat of fusion of ice ; Specific heat of water ]
HARD
A graph of vapour pressure and temperature for three different liquids and is shown below:
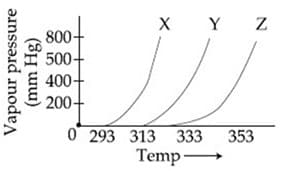
The following inferences are made:
has higher intermolecular interactions compared to
has lower intermolecular interactions compared to
has lower intermolecular interactions compared to
The correct inferences is/are:
MEDIUM
A liquid does not wet the solid surface if angle of contact is:
MEDIUM
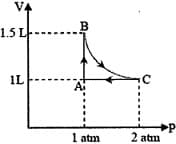
A system consisting of 1 mol of an ideal gas undergoes a reversible process, (schematically indicated in the figure below). If the temperature at the starting point A is 300 K and the work done in the process is 1 L atm, the heat exchanged in the entire process in L atm is
EASY
The liquid having maximum surface tension is
EASY
The work done during combustion of of ethane, at is (Given , atomic mass )
HARD
A reversible cyclic process for an ideal gas is shown below. Here, are pressure, volume and temperature, respectively. The thermodynamic parameters are heat, work, enthalpy and internal energy, respectively.
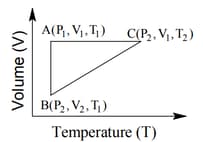
The correct option(s) is (are)
EASY
Calculate the work done during compression of mol of an ideal gas from a volume of to at against a pressure of .
EASY
An ideal gas undergoes isothermal expansion at constant pressure. During the process:
EASY
A gas undergoes change from state to state . In this process, the heat absorbed and work done by the gas is , respectively. Now gas is brought back to by another process during which of heat is evolved. In this reverse process of .
MEDIUM
Based on the given figure, the number of correct statement/s is/are
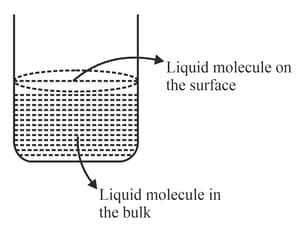
A. Surface tension is the outcome of equal attractive and repulsion forces acting on the liquid molecule in bulk.
B. Surface tension is due to uneven forces acting on the molecules present on the surface.
C. The molecule in the bulk can never come to the liquid surface.
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system.
EASY
The viscosity of a liquid can be decreased by
EASY
The unit of coefficient of viscosity is:

