HARD
JEE Advanced
IMPORTANT
Earn 100
The plot given below shows curves (where, is the pressure and is the temperature) for two solvents and and isomolal solutions of in these solvents. completely dissociates in both the solvents.
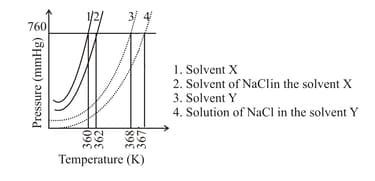
On the addition of equal number of moles of a non-volatile solute in equal amount (in ) of these solvents, the elevation of boiling point of solvent is three times that of solvent . The solute is known to undergo dimerization in these solvents. If the degree of dimerization is in the solvent , the degree of dimerization in the solvent is _______.

On the addition of equal number of moles of a non-volatile solute in equal amount (in ) of these solvents, the elevation of boiling point of solvent is three times that of solvent . The solute is known to undergo dimerization in these solvents. If the degree of dimerization is in the solvent , the degree of dimerization in the solvent is _______.
38.33% studentsanswered this correctly
Important Questions on Solutions
MEDIUM
JEE Advanced
IMPORTANT
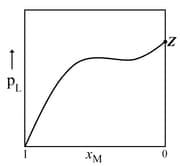
For a solution formed by mixing liquids and , the vapour pressure of plotted against the mole fraction of in solution is shown in the following figure, Here, and represent mole fractions of and , respectively, in the solution. The correct statement(s) applicable to this system is(are) -
HARD
JEE Advanced
IMPORTANT
Benzene and naphthalene form an ideal solution at room temperature. For this process, the true statement(s) is (are)
