EASY
NEET
IMPORTANT
Earn 100
The temperature of the system decreases in an:
(a)Adiabatic compression
(b)Isothermal compression
(c)Isothermal expansion
(d)Adiabatic expansion
50% studentsanswered this correctly

Important Questions on Thermodynamics
MEDIUM
NEET
IMPORTANT
HARD
NEET
IMPORTANT
HARD
NEET
IMPORTANT
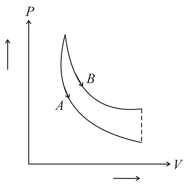
In figure, and are two adiabatic curves for two different gases. Then, and correspond to,
EASY
NEET
IMPORTANT
EASY
NEET
IMPORTANT
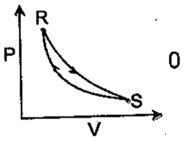
Consider the cyclic process as shown in the Fig. You are toid that one of the path is adiabatic and the other one isothermal. Which one of the following is(are) true?
EASY
NEET
IMPORTANT
MEDIUM
NEET
IMPORTANT
Predict which of the following reaction has a positive entropy change?
MEDIUM
NEET
IMPORTANT
Which of the following reactions is associated with the most negative change in entropy?
