What is the cell reaction occurring in Daniel cell (Galvanic cell)?
Important Questions on Electrochemistry
The following reaction takes place at in an electrochemical cell involving two metals and ,
with and in the respective half-cells, the cell EMF is . The equilibrium constant of the reaction is closest to;
Represent a cell consisting of | half cell and | half cell and write the cell reaction.
For the cell , when the concentration of is times the concentration of , the expression for is
Faraday's constant, universal gas constant, temperature,
At , the of the galvanic cell mentioned below is
Depict the galvanic cell in which the reaction takes place. Further show, which of the electrode is negatively charged.
.
In the given figure the experiment of conductivity solution is shown. Label against and .
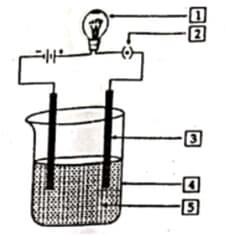
Daniell cell converts the chemical energy liberated during the redox reaction to electrical energy.
Identify the anode and cathode in Daniell cell.
,
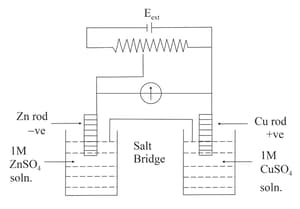
Identify the incorrect statement from the options below for the above cell:

