HARD
Earn 100
What will happen when we apply pressure and compress a gas enclosed in a cylinder? Will the particles come closer?
Important Questions on Matter in our Surroundings
MEDIUM
When does ice melt at the same temperature (273 Kelvin), which one of the following has more energy?
MEDIUM
MEDIUM
HARD
MEDIUM
MEDIUM
HARD
MEDIUM
HARD
EASY
EASY
EASY
(Latent heat of ice is and )
MEDIUM
MEDIUM
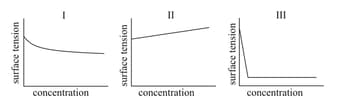
HARD
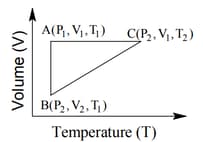
The correct option(s) is (are)
MEDIUM
The combination of plots which does not represent isothermal expansion of an ideal gas is
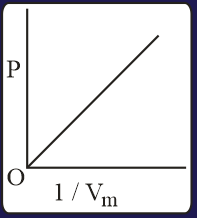
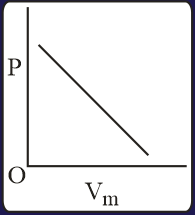
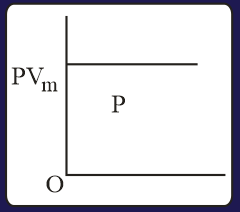
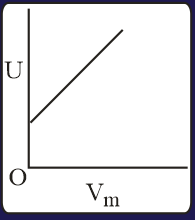
MEDIUM
HARD
HARD
