HARD
JEE Main
IMPORTANT
Earn 100
When a plot of boiling point versus composition is plotted, then the type of graph that cannot be obtained is:
(a)
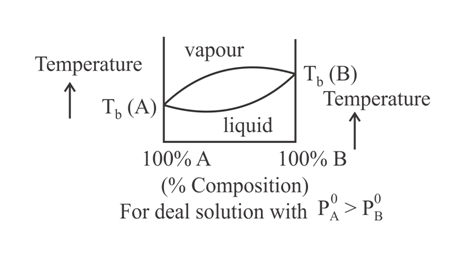
(b)
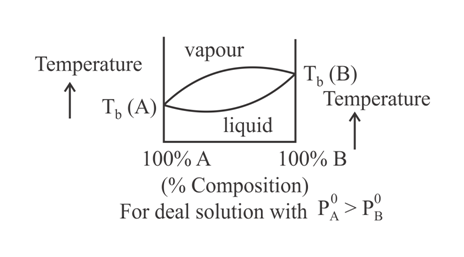
(c)
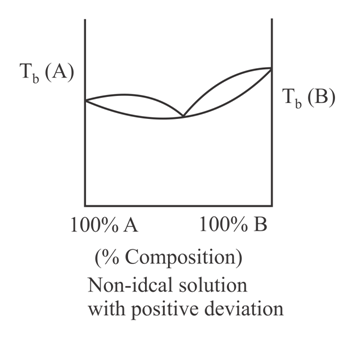
(d)
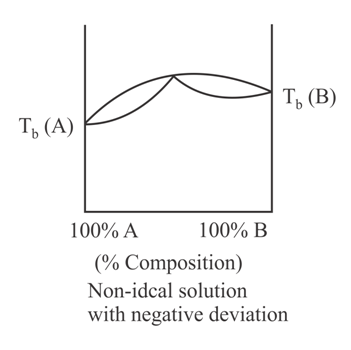
50% studentsanswered this correctly

Important Questions on Solutions
MEDIUM
JEE Main
IMPORTANT
Which of the following graphs represents the behavior of ideal binary liquid mixture?
(Here, and are the mole fractions of components and in vapour.)
MEDIUM
JEE Main
IMPORTANT
On mixing mole of and mole of , one can conclude
MEDIUM
JEE Main
IMPORTANT
Benzene and naphthalene form an ideal solution at room temperature. For this process, the true statement(s) is (are)
MEDIUM
JEE Main
IMPORTANT
In the following statements, which is/are false for a solution of chloroform and acetone?
MEDIUM
JEE Main
IMPORTANT
Composition of an azeotrope
HARD
JEE Main
IMPORTANT
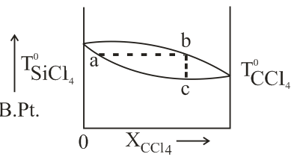
The diagram given below depicts the boiling point as the function of composition of the mixture of and . Which of the following statement about the diagram is/are true?
MEDIUM
JEE Main
IMPORTANT
Explain why on the addition of mole of to of water, the boiling point of water increases, while the addition of a mole of methyl alcohol to of water, decreases its boiling point?
EASY
JEE Main
IMPORTANT
What is the reason that the vapour pressure of an aqueous solution of glucose is lower than that of water?
