EASY
Earn 100
Which is not true in respect of beryllium chemistry?
(a)Beryllium is amphoteric
(b)It forms unusual carbide
(c) is amphoteric
(d)Beryllium halides are electron deficient
50% studentsanswered this correctly
Important Questions on The s-Block Elements
EASY
EASY
EASY
EASY
EASY
MEDIUM
MEDIUM
Given below are two statements:
Statement I : The chlorides of and have Cl-bridged structure. Both are soluble in organic solvents and act as Lewis bases.
Statement II: Hydroxides of and dissolve in excess alkali to give beryllate and aluminate ions. In the light of the above statements. Choose the correct answer from the options given below.
MEDIUM
MEDIUM
EASY
Among the statement (I – IV), the correct ones are:
(I) Be has smaller atomic radius compared to Mg.
(II) Be has higher ionization enthalpy than AI.
(III) Charge/radius ratio of Be is greater than that of Al.
(IV) Both Be and Al form mainly covalent compounds
EASY
HARD
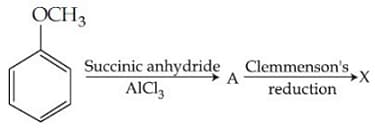
X is :
MEDIUM
MEDIUM
MEDIUM
MEDIUM
HARD
EASY
Reason (R) : Both and have almost same ionic radius
The correct option among the following is
EASY
EASY

