EASY
JEE Advanced
IMPORTANT
Earn 100
Why sodium is kept under kerosene?

Important Questions on s-Block Group 1 (Alkali Metals)
EASY
JEE Advanced
IMPORTANT
Alkali metals are obtained by the electrolysis of the molten salts and not by the electrolysis of their aqueous solutions.
EASY
JEE Advanced
IMPORTANT
Melting point and boiling point of alkali metals decrease from to . Explain.
EASY
JEE Advanced
IMPORTANT
Lithium hydroxide has been used by astronauts for a particular purpose, explain it with reaction.
MEDIUM
JEE Advanced
IMPORTANT
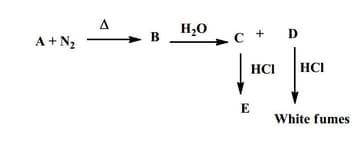
Identify (alkali metal) to .
EASY
JEE Advanced
IMPORTANT
Identify to
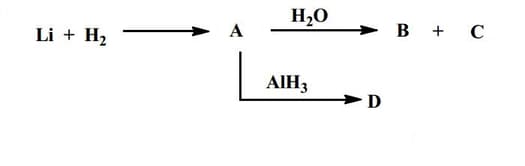
EASY
JEE Advanced
IMPORTANT
Identify and .
Both and decolourize acidified
EASY
JEE Advanced
IMPORTANT
A metal (A) is silver white and soft. It remains unchanged in dry air but is readily oxidized by moist air becoming covered with a blue film. Metal chloride solution gives yellow (B) with sodium hexanitrocobaltate (III) solution. Similar test is given by ammonium ion also. On adding perchloric acid white crystalline . (C) is formed which is slightly soluble in water but practically insoluble in absolute alcohol. Ammonium ion does not respond in this test. Identify (A), (B) and (C) and write balanced chemical reactions also.
MEDIUM
JEE Advanced
IMPORTANT
Why does the following reaction:
Proceed better with than with ?