In the given figure an ideal gas changes its state from to state by two paths and .
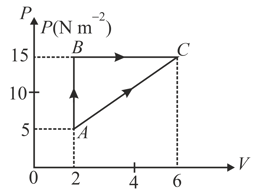
(i) The internal energy of gas at is and amount of heat supplied to change its state to through the path is . Calculate the internal energy at .
(ii) The internal energy of gas at is . Find the amount of heat supplied to the gas from to .


Important Questions on Thermodynamics
(i) If work done by the gas is then find the value of .
(ii) If increase in internal energy is then find the value of .
(iii) If amount of heat supplied is then find the value of .
(iv) If molar specific heat of the gas is then find the value of .
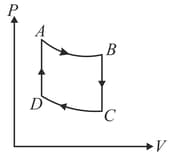
: Adiabatic expansion : Cooling at constant volume
: Adiabatic compression
: Heating at constant volume
The pressure and temperature at , etc., are denoted by etc., respectively. Given that , and .
Calculate the following quantities : (i) The work done by the gas in the process
(ii) The heat lost by the gas in the process
(iii) The temperature . (Given : )
At two moles of an ideal monoatomic gas occupy a volume . The gas expands adiabatically to a volume . Calculate:
(i) the final temperature of the gas,
(ii) change in its internal energy and
(iii) the work done by the gas during the process.
(a) Find the value of .
(b) What is the efficiency of the cycle?
