HARD
JEE Main
IMPORTANT
Earn 100
The slope of vs was made against insulin concentration in and temperature . If is in and the slope of line obtained is , which of the following are not the molar mass (in ) of insulin?
(a)
(b)
(c)
(d)
50% studentsanswered this correctly

Important Questions on Solutions
MEDIUM
JEE Main
IMPORTANT
A graph showing the variation of osmotic pressure versus molar concentration of an aqueous solution at temperature is given below.
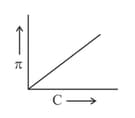
The slope of the line doesn't represent
MEDIUM
JEE Main
IMPORTANT
Calculate the osmotic pressure of (mass/volume anhydrous) solution at assuming ionisation in .
EASY
JEE Main
IMPORTANT
Under what conditions Van't Hoff factor is equal to unity?
EASY
JEE Main
IMPORTANT
Under what conditions van't Hoff factor is less than ?
EASY
JEE Main
IMPORTANT
Under what conditions van't Hoff factor is greater than ? Explain your answer.
MEDIUM
JEE Main
IMPORTANT
If the solution taken is too concentrated, then how will it affect the molar mass of the solute?
HARD
JEE Main
IMPORTANT
A dilute solution contains mol of solute in of a solvent with molal elevation constant the solute dimerises in solution as .
Show that the equilibrium constant for this dimer formation is , where is the elevation in boiling point for the given solution.
MEDIUM
JEE Main
IMPORTANT
The van't Hoff factor for a compound which undergoes dissociation in one solvent and association in other solvent is respectively
