Second Law of Thermodynamics
Second Law of Thermodynamics: Overview
This topic covers concepts such as Second Law of Thermodynamics, Terms in Second Law of Thermodynamics, Kelvin-Planck Statement, and Clausius Statement.
Important Questions on Second Law of Thermodynamics
An engine operating between the boiling and freezing points of water will have
A. Efficiency more than .
B. Efficiency less than the efficiency of a Carnot engine operating between the same two temperatures.
C. Efficiency equal to .
D. Efficiency less than .
Choose the correct answer from the options given below
"Heat cannot be itself flow from a body at lower temperature to a body at higher temperature". This statement corresponds to
No process is possible whose sole result is the transfer of heat from a colder object to a hotter object. This is Clausius statement for
The second law of thermodynamics may be written is used symbols as-
The heat taken from a sink is heated on-
The _____ of the thermodynamics states that, no process is possibel whose sole results is the absorption heat from a reservation and the complete conversion of heat into work.
_________ of thermodynamics is used for understanding the concept of entropy change.
PMM2 is the machine which violates-
For an increase in the entropy define ____ of nature.
Entropy is the third law of thermodynamics.
Which of the following option are correct for the second law of thermodynamics?
What is the hypothetical example of the Kelvin – Planck Statement?
Kelvin Planck Statement deals with conversion of work into _____, conservation of work, conservation of heat, and conversion of heat into work.
Efficiency of a engine always less than unity.
Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
Mention the causes for irreversibility of a process.
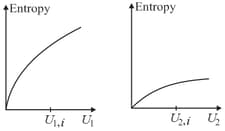
Graphs below show the entropy versus energy of two systems and at constant volume. The initial energies of the systems are indicated by and respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.
Clausius's statement is a part of
