Second Law of Thermodynamics
Second Law of Thermodynamics: Overview
This topic covers concepts, such as, Second Law of Thermodynamics, Kelvin-Planck Statement & Clausius Statement etc.
Important Questions on Second Law of Thermodynamics
What is the entropy change (in ) when one mole of ice is converted into water at ? (The enthalpy change for the conversion of ice to liquid water )
According to second law of thermodynamics :
What is the Kelvin Planck Statement
An ideal gas heat engine operates in Carnot cycle between and . It absorbs cal of heat at high temperature. Amount of heat converted to work is:
Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
State Clausius statement of second law of thermodynamics.
State Kelvin-Planck statement of the second law of thermodynamics
Write the statement of second law of thermodynamics
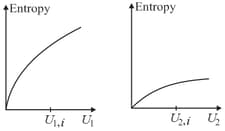
Graphs below show the entropy versus energy of two systems and at constant volume. The initial energies of the systems are indicated by and respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.
The change in entropy when a ice cube at is transformed into water at in is
Take ,
Clausius's statement is a part of
What do you mean by Clausius's statement?
The wavelength associated with the electron will be
Which of the following statements is true? The entropy of the universe
The change in the entropy of a 1 mole of an ideal gas which went through an isothermal process from an initial state to the final state is equal to
An ideal gas heat engine operates in Carnot cycle between and .It absorbs cal of heat at high temperature. Amount of heat converted to work is :
Work done by an engine is when a heat engine absorbs heat at temperature and heat at temperature. This data:
"Heat cannot by itself flow from a body at lower temperature to a body at higher temperature" is a statement or consequence of
