Orbital Overlap
Orbital Overlap: Overview
This Topic covers sub-topics such as Overlapping of Atomic Orbitals, Axial Overlapping, Lateral Overlapping, Formation of Sigma and Pi bonds in Ethene, Formation of Sigma and Pi bonds in Ethyne and, Comparison of Sigma and Pi Bonds
Important Questions on Orbital Overlap
Propyne molecule contains sigma and _____ pi bonds.
The sum of number of sigma and pi bonds formed between two carbon atoms in are:
The number of and bonds in allyl isocyanide are
bonding is not present in the molecule :
The combination of orbital that cannot produce non-bonding molecular orbital is (if an inter-nuclear axis is -axis):
If orbital of and orbital of from bond along a particular molecular axis, then which bond will be formed along same molecular axis by combination of orbitals of and atom.
Which types of bonds are present between two carbon atoms in acetylene molecule?
Select the correct order of bond angle of the following species.
Which of the following overlap is correct?
In the group 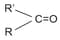 , the carbonyl carbon is joined to other atoms by
, the carbonyl carbon is joined to other atoms by
Which of the following ions does not involve bonding ?
Nitrogen forms , but phosphorus is converted into from . What is the reason for it?
A molecule () has
(i) four sigma bonds formed by the overlap of and orbitals
(ii) one sigma bond formed by and orbitals and
(iii) one bond formed by and orbitals. Which of the following is ?
Compound X is anhydride of sulphuric acid. The number of the number of - bonds present in X are, respectively.
In acetylene molecule, What is between the carbon atoms?
The number of and - bonds in a molecule of acetonitrile are respectively,
Where is the nodal plane in the π-bond of ethene is located?
The nodal plane in the - bond of ethene is located
